Check out our Frequently Asked Questions
![]() Find a list of our past PIP/EHRIP email communications at our Archived Announcements
Find a list of our past PIP/EHRIP email communications at our Archived Announcements
 MAPIR is accepting PY2021 applications through July 31, 2021.
MAPIR is accepting PY2021 applications through July 31, 2021.
Sign up to receive email updates: ahs.dvhaEHRIP@vermont.gov
The PY2021 Attestation Window is Open through July 31, 2021 (posted 11/18/2022)
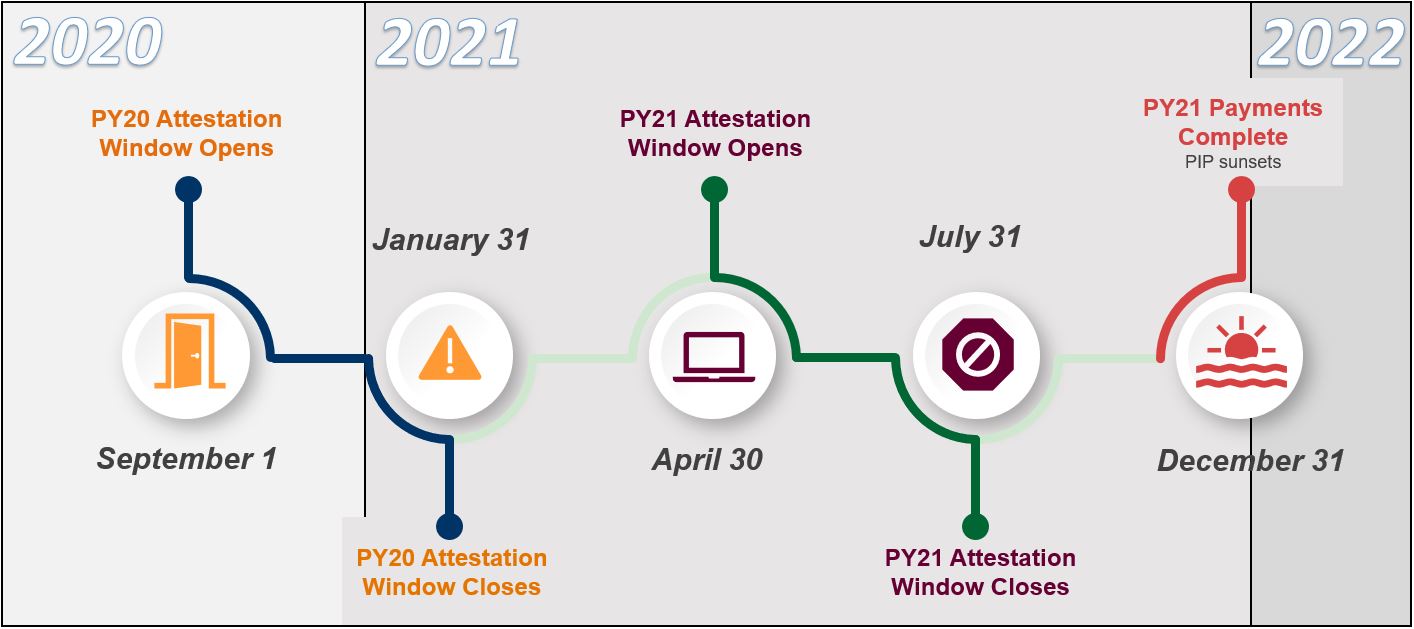
PIP Timeline:
The Electronic Health Record Incentive Payment program was established by the 2009 Health Information Technology for Economic and Clinical Health (HITECH) Act of the American Recovery & Reinvestment Act (ARRA). The program is designed to support providers during the period of health information technology transition and to improve the quality, safety and efficiency of patient health care through the use of electronic health records (EHRs).
CMS continues to align and streamline their Quality Reporting initiatives to move beyond the early requirements of Meaningful Use (MU), to a new phase of EHR measurement focused on interoperability and improving patient access to health information.
To better reflect this focus, CMS renamed the Medicaid 'EHR Incentive Program’ to the 'Promoting Interoperability Program' for eligible hospitals, critical access hospitals, and Medicaid eligible professionals. As terminology is updated across websites and communications, you will see references to "Promoting Interoperability/EHRIP or "PIP/EHRIP" guidance.
The PIP/EHRIP will provide incentive payments to eligible professionals, eligible hospitals and critical access hospitals as they continue demonstrate meaningful use of certified EHR technology. Eligible providers will be able to apply for PIP/EHRIP payments through 2021.
CLOSING TIME (posted 07/22/2021)
The final week to attest for the final program year of the Vermont Medicaid Promoting Interoperability Program is here. All applications must be submitted by July 31, 2021. If there is any reason why this cannot be accomplished, please reach out to your PIP Team for assistance: ahs.DVHAehrip@vermont.gov
SRA REMINDER (posted 07/22/2021)
As a reminder, the requirement to complete a Security Risk Assessment (SRA) has been modified for PY2021. Previously, a provider was required to attest to an SRA being completed during the program year for which they are attesting; the SRA had to already be completed prior to attestation and provide documentation of the SRA with the application.
When providers attest for PY2021, they will be asked if the measure (completing an SRA) has been completed prior to the date of attestation. If the response is "no", then they will be asked to attest that the SRA will be completed no later than December 31, 2021 and that they understand that their incentive payment will be subject to recoupment for failure to do so. If the response is “yes,” the preparer should upload the 2021 SRA with the application in MAPIR.
All attesting providers must upload a copy of their 2021 SRA to MAPIR by January 31, 2022, otherwise they will automatically be selected for audit.
HELP IMPROVE THE SRA (posted 07/22/2021)
The HHS Office for Civil Rights and the HHS Office of the National Coordinator for Health Information Technology (ONC) has created several iterations of a Security Risk Assessment tool. They are seeking user feedback and improvement suggestions. The SRA Tool is designed to help small and medium-sized healthcare providers conduct a security risk assessment, as required by the HIPAA Security Rule and the Centers for Medicare and Medicaid Services (CMS) Promoting Interoperability Program. If you have suggestions on how to improve the Tool, OCR asks you to complete their short survey by July 31, 2021.
MAPIR ACCESS TROUBLESHOOTING (posted 07/22/2021)
As PY2021 applications have been started in MAPIR, we’ve been receiving more questions about logging into MAPIR and how to
troubleshoot issues that might arise. Here are some tips:
- MAPIR is accessed through the Vermont Medicaid Portal. The User ID for the Provider Portal will be the same User ID as for the Provider Management Module.
- If you do not have a User ID, fill out a Trading Partner Agreement and an EDI Registration Form. Send these forms to vtedicoordinator@dxc.com.
- If you have forgotten the User ID or if you have questions about the forms above, please email vtedicoordinator@dxc.com.
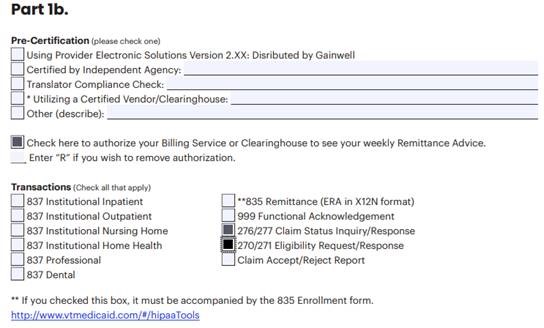
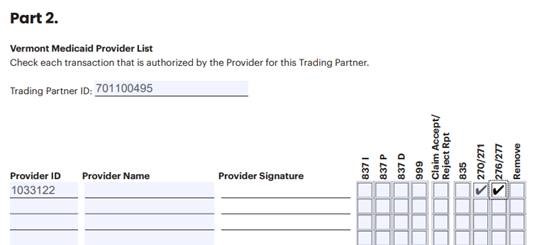
Part 1b and Part 2 of the EDI Registration Form should be filled out to look like this:
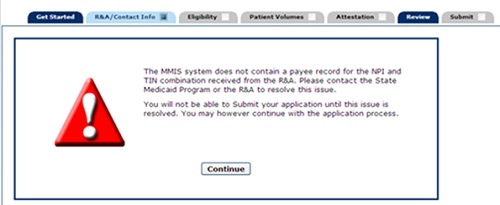
- If you see the error below, you will need to go to the CMS R&A site and check the registration for the provider to ensure that the "Payee TIN Type" on the provider's registration was set to "Group Reassignment." If "Group Reassignment" was not selected, you can go back in and modify the Payee TIN Type to "Group Reassignment." Complete all screens on this site and submit. It may take 24-48 hours for changes to be reflected in MAPIR.
For assistance with the R&A site, email NLRProdSupport@cms.hhs.gov
For additional guidance accessing MAPIR, please visit this webpage and check out our MAPIR User Guides.
ATTESTATION FAQS (posted 06/25/2021)
As more providers are attesting for PY2021 in MAPIR, some commonly-occurring questions are coming into our help desk email. Some of these questions and their answers are below.
We are trying to attest for a provider, but we see in the CMS Registration and Attestation site that he is registered for Medicare, not Medicaid. The CMS R&A is not allowing me to switch the program affiliation to Medicaid. Can we do this? No. The last year for a provider to switch from the Medicare to Medicaid incentive program was before the application deadline for submissions for Program Year 2014. Going forward, a provider must have received at least one Medicaid PIP/EHRIP payment in order to be qualified to participate in the program.
One of our eligible providers left our employment and went to another practice in May 2021. The provider’s new practice has taken him on as an Eligible Professional. Can we still submit a MAPIR application for him for Program Year 2021? No. Eligibility for PIP is provider-based, not practice-based. An Eligible Professional who leaves a practice retains their eligibility to participate in the PIP with their new practice or jurisdiction. Unless there is a specific authorized agreement between the provider and your practice for a Program Year 2021 PIP payment, you will not submit an application in MAPIR on behalf of this provider.
We have a provider who is failing one Meaningful Use measure threshold. Patient volume and all other Objectives and Measures are being met. Do we complete an application in MAPIR anyway? No. Meaningful Use is an all-or-nothing construct. You may try a different 90-day period to see if they meet the measure. If the provider does not meet the threshold for one of the objectives or measures, they will not meet the requirements for Meaningful Use, and they will not submit an application in MAPIR.
I have completed a provider’s attestation and submitted it in MAPIR. But I then discovered an error in the data I entered. I can’t access the application to correct it. What should I do? The PIP Team can put the application back into “Incomplete” status so you can correct the data and resubmit the application. Just send an email to: ahs.dvhaEHRIP@vermont.gov
For additional FAQs from CMS regarding the Promoting Interoperability Program click here.
REQUIRED DOCUMENTATION FOR ATTESTATION (posted 06/11/2021)
When attesting to Meaningful Use in MAPIR, supporting documentation for some objectives and measures is required. Other documentation is recommended and may be required upon request from the PIP team.
Required at the time of attestation:
- Group Definition file if attesting using the group patient volume methodology
- Security Risk Analysis (SRA) completed during the applicable program year (see "IMPORTANT SRA UPDATE" below)
- MU report from your Electronic Health Record (EHR) system supporting attestation that include numerators, denominators, exclusions, and exception when applicable. To avoid delays in processing, please double-check these entries.
- Clinical Quality Measure (CQM) report from your EHR.
- Documentation to support active engagement or exclusion taken for Objective 8, options 4a & b, and 5a &b. For more information about Public Health Objective 8, see our website.
Recommended: (These may be requested after application submission and are mandatory in the event of an audit)
- Patient Volume data file
- Proof of CEHRT
IMPORTANT SRA UPDATE (posted 06/11/2021)
When providers attest for PY2021, they will be asked if the measure (completing/reviewing an SRA) has been completed prior to the date of attestation.
If the response is "no", then they will be asked to attest that the SRA/review will be completed no later than December 31, 2021 and that they understand that their incentive payment will be subject to recoupment for failure to do so.
If the response is "yes", then the preparer must upload the SRA into MAPIR at the time of attestation.
All attesting providers must upload a copy of their 2021 SRA/review to MAPIR by January 31, 2022, otherwise they will automatically be selected for audit.
PROVIDER MEDICAID STATUS (posted 5/28/2021)
An important reminder that if an eligible professional’s Vermont Medicaid enrollment lapses at any time after an application is started and before an incentive payment is received, the application will automatically abort from the MAPIR system. The attestation must then be restarted from the beginning in MAPIR after the EP becomes fully re-enrolled in Vermont Medicaid. In the case of PY2021, all incentive payments will be made by December 31, 2021.
ALIGNING PROVIDER INFO AT THE R&A SITE (posted 5/28/2021)
Now that MAPIR is available to accept applications for Program Year 2021, there are a number of recurring questions we receive about the “R&A Verification” screen. The information on this screen is populated from the data feed that is sent directly from CMS. The fields on this screen are not interactive; you cannot alter the information that is displaying. You can only answer “Yes” or “No” to the question “Is this information accurate?” at the bottom of the screen.
If you answer “No,” MAPIR will warn that the information needs to be corrected at the CMS R&A site. You will NOT be able to move past this screen as long as the “No” is selected.
If any information on the screen requires correction (EXCEPT for the CMS EHR Certification Number explained below), you will need to access the provider’s information at the CMS R&A site to ensure that the NPI, Payee NPI, Payee TIN, contact email, etc. is accurate. Please note that It will take 24 to 48 hours for edits to propagate through from the CMS system to the MAPIR system.
If a new, updated CEHRT ID (different than the one displaying on this screen) was entered in order to start this application, it will be saved and will automatically update the CMS R&A site information when the application is submitted and processed.
If all other information on this screen is correct except for the CMS EHR Certification Number, you may choose the “Yes” button and “Save & Continue” the provider is attesting to using a new CEHRT ID in Program Year 2021, due to a system upgrade or vendor switch, you DO NOT have exit this screen to update the CEHRT ID at the CMS R&A site. If all other information on this screen is correct except for the CMS EHR Certification Number, you may choose the “Yes” button and “Save & Continue.”
BROADBAND IN VERMONT (posted 5/17/2021)
While it’s no secret that access to fast and reliable internet is a not a given in Vermont, the lack of high-quality internet service is currently in the spotlight as broadband expansion bills are being moved through the Vermont legislature. Part of the windfall COVID-19 funds from Washington is very likely going towards improving broadband availability in our state. This is related to the Promoting Interoperability Program because Objectives 5, 6, and 7 all include exclusions for some measures that involve the availability of broadband internet access in the county where an EP practices. The exclusion is as follows:
“An EP may take an exclusion if… They conduct 50 percent or more of their patient encounters in a county that does not have 50 percent or more of its housing units with 4Mbps broadband availability according to the latest information available from the Federal Communications Commission (FCC) on the first day of the EHR reporting period."
It is easy to bemoan the current state of broadband internet in Vermont, however any EP with at least 50% of their encounters in Vermont does not qualify for these exclusions. According to the FCC and Vermont Department of Public Service, there are no counties that meet the requirements of the broadband exclusions for Objectives 5, 6, and 7.
IT'S NOT TOO LATE FOR PY2021 (posted 5/17/2021)
You may think that because we are in the attestation window for PY2021 that it’s too late for you to attest to meaningful use for this year. However, there is still time to be successful in leveraging that incentive payment! Both the MU attestation period and eCQM periods are only 90 days. The latest these reporting periods could start is May 1, 2021. There's still time!
As soon as possible, consider the following tasks:
- Email ahs.dvhaEHRIP@vermont.gov to request verification of eligibility for providers at your organization or request a consultation.
- Visit https://healthdata.vermont.gov/ehrip/PY2021 to review PY2021 requirements.
- Examine your current EHR MU reports. Where can improvements be made?
- Plan and implement any operational changes that are necessary to meet MU before May 1st.
- Visit https://healthdata.vermont.gov/ehrip/apply to make sure you’re ready for attestation and have the MAPIR User Guides handy.
The attestation window closes July 31, 2021
PY2021 ATTESTATION WINDOW IS OPEN - NOW WHAT?! (posted 4/30/2021)
The attestation window is open for PY2021 and it’s time to attest for the EP’s in your organization! Here is a quick to-do-list to get you off on the right foot:
- Assess Patient Volume
- Determine if the EP(s) will be using individual or group patient volume to meet the 30% Medicaid requirement.
- Choose 90 day period where this requirement is met and prepare supporting documentation
- Print/download the MAPIR 6.4 User Guides
- If you have a second monitor at your work-station, it is recommended that you have the PDF open on one and MAPIR on the other. Alternatively, you may want to print portions of these user guides, especially Part 2C and Part 3
- Print/download the Public Health Documentation Guides (VT and NH)
- Specifically you may want to print the guides for Options 4 (Public Health) and 5 (Clinical Registry) because these objectives require specific documentation for both attestation AND exclusions that will need EP signature(s).
- Prepare supporting documentation
- Meaningful Use Reports
- Patient Volume Report
- Security Risk Analysis Documentation (NEW requirements here)
- Log in to MAPIR
- Access to MAPIR is found through the Vermont Medicaid Portal. For more instruction, see MAPIR User Guide Part 1.
Still need help? Email us at ahs.DVHAehrip@vermont.gov or access the MAPIR FAQ’s here. If you need to check eligibility of any providers, please email us their names and NPI numbers.
FINAL PROGRAM YEAR TIMELINE (posted 4/30/2021)
UPDATED MAPIR USER GUIDES AVAILABLEI (posted 4/15/2021)
Have you ever utilized our MAPIR User Guides during the attestation process? If not, you’re missing out! These four PDF’s offer valuable step-by-step guidance and screenshots of every single page in MAPIR that you’ll encounter. From logging in to MAPIR through uploading documents after submission, explanations of errors messages to requirements of your internet browser are all addressed.
It is highly recommended that preparers have these PDF’s handy during the attestation process; it may be quite helpful to follow along page-by-page as you work through the application in MAPIR as common challenges that arise at each page are addressed along the way.
These user guides are updated with each version of MAPIR. MAPIR 6.4 is in use for PY2021 attestations; if you downloaded previous versions, please make sure to replace them with the latest version for 6.4. User guides can be found on our website here.
REMINDER: NEW SRA REQUIREMENT (posted 4/15/2021)
A reminder that the Security Risk Assessment requirement has been updated for PY2021:
When providers attest for PY2021, they will be asked if the measure (completing an SRA) has been completed prior to the date of attestation. If the response is "no", then they will be asked to attest that the SRA will be completed no later than December 31, 2021 and that they understand that their incentive payment will be subject to recoupment for failure to do so. SRA documentation must be uploaded into MAPIR by January 31, 2022; failure to do so will result in the application being selected for audit.
If you would like assistance performing your SRA, there are resources available! Click here to visit our website, or reach out to Mary Donati at VITL for free SRA assistance by emailing her at mdonati@vitl.net. VITL offers SRA services free-of-charge, but also offers a variety of services at additional costs.
PY2021 eCQM GUIDES ARE LIVE (posted 4/15/2021)
Guidance for reporting CQMs for PY2021 has been updated and added to our PY2021 webpage.
Check out this PDF for an overview of requirements. If a deeper dive, including screenshots of each CQM screen in MAPIR is helpful, this PDF will provide additional information.
PIP DOESN'T END WITH 2021... (posted 4/2/2021)
All program participants should be aware that audit activity for the Vermont Medicaid Promoting Interoperability Program will continue into 2023.
Though incentive payments will not extend beyond 2021, it is vital that all participants keep all supporting documentation through 2023 in the case of a post-payment audit.
For more information about audit activity, visit our website.
PATIENT VOLUME: 101 (posted 4/2/2021)
Since its inception, the Medicaid Promoting Interoperability Program has a Medicaid patient volume (PV) requirement that must be demonstrated for each year of program participation.
Definition: A Medicaid encounter is any service rendered to an individual on any one day where:
- Medicaid paid for part or all of the service, OR
- Medicaid paid all or part of the individual's premiums, co-payments, and cost-sharing, OR
- The individual was enrolled in a Medicaid program at the time the billable service was provided.
The Requirements:
- For all EP types (minus pediatricians): 30% of all encounters during the PV reporting period must be Medicaid encounters
- For all pediatricians: 20% of all encounters during the PV reporting period must be Medicaid encounters
- For those practicing at an FQHC: 30% of all encounters during the PV reporting period must be Medicaid encounters and/or Needy encounters. For Needy encounters, services rendered on any one day to an individual can be counted if:
- The services were furnished at no cost; OR
- The services were paid for at a reduced cost based on a sliding scale determined by the individual's ability to pay.
These volumes must be met for a 90-day period. There are two options for selecting this period:
- Calendar year preceding the program year
- 12 months preceding the attestation date
Please note that when attesting for PY2021, the PV reporting period may not overlap with any PV reporting period used for a previous attestation.
Individual vs. Group:
An EP may elect to use a clinic or group practice’s PV as a proxy for their own under three conditions:
- The clinic or group practice's PV is appropriate as a PV methodology calculation for the EP (for example, if an EP only sees Medicare, commercial, or self-pay patients, this is not an appropriate calculation); AND
- There is an auditable data source to support the clinic's PV determination; AND
- The practice and EP’s use one methodology in each year (all attesting EP’s from a group must either uses individual PV methodology OR group PV methodology)
Watch Out: Don’t count multiple claims for services for the same patient by the same provider on the same day! PV requirements assess encounters, not claims. The EP should remove duplicate encounters on the same day when calculating PV.However, services rendered to the same patient by different providers on the same day are allowed to be counted discretely by each provider.
Required and Recommended Documentation:
If attesting using group PV, a Group Definition file must be submitted with the application in MAPIR.The Group Definition should include a list of all provider names in the group (regardless of whether they are attesting for MU) and their individual billing NPI numbers.
Regardless of PV methodology, a PV data file to support PV attestation is highly recommended. It may be requested after application submission if not initially uploaded into MAPIR. Be aware: this data file is required
in the event of an audit.
For additional information including Patient Volume Tools and Rule References, please visit our website.
WHO ARE YOUR ELIGIBLE PROVIDERS? (posted 3/19/2021)
In preparing for PY2021 Attestation, it is best practice to review the staff at your organization to assess eligibility for a PIP incentive payment. Follow this workflow below to help you determine eligibility.
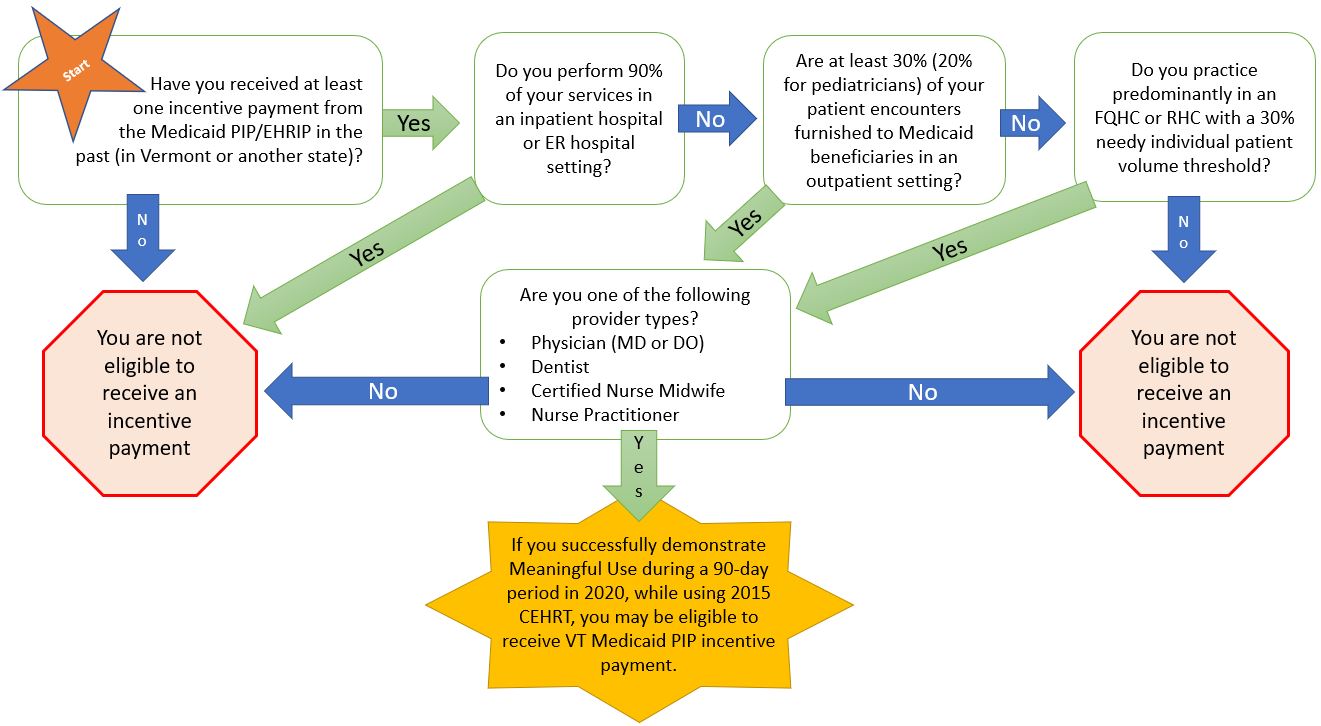
Still not sure if someone if eligible? Send us a list of provider names and NPI numbers at ahs.DVHAehrip@vermont.gov and we will verify eligibility.
Interested in attesting to our program, but don’t know where to start? We are here to help walk you through the whole process. Email us at ahs.DVHAehrip@vermont.gov and request a webinar. Depending on the level of knowledge regarding PIP andPY2021, the presentation will be customized to suit what exactly you need to know to successfully attest in PY2021. Schedule one TODAY before the attestation window opens April 30th.
PIP/EHRIP WEBSITE UPDATED (posted 3/19/2021)
Our website has been updated with PY2021-specific information. Public health documentation aids, audit information, and program requirements are all live. Stay tuned for updated MAPIR User Guides in mid-April.
PY2021 LAST REPORTING PERIOD (posted 3/19/2021)
The last recommended 90-day MU/EHR period of 2021 starts April 26, 2021. This will allow for 5 business days to complete applications in MAPIR before the window closes on July 31st.
SRA’s: FROM WHY TO HOW (posted 3/4/2021)
Today many patients’ protected health information is stored electronically, so the risk of a breach of their ePHI, or electronic protected health information, is very real. By conducting regular assessments and addressing any risks, health care organizations can help to ensure that ePHI is kept secure.
Conducting or reviewing a security risk analysis (SRA) to meet the standards of Health Insurance Portability and Accountability Act of 1996 (HIPAA) Security Rule is included in the meaningful use requirements of the Medicaid PIP/EHR Incentive Program every year.
PIP Attestation Requirements: Conducting an SRA was required when certified EHR technology was adopted in the first reporting year. In subsequent years or when changes to the practice or electronic systems occur, a review must be conducted. Any security updates and deficiencies that are identified in the review should be included in the provider’s risk management process and implemented or corrected as dictated by that process.
The SRA must assess the potential risks and vulnerabilities to the confidentiality, availability, and integrity of all ePHI that an organization creates, receives, maintains, or transmits. This includes ePHI in all forms of electronic media, such as hard drives, floppy disks, CDs, DVDs, smart cards or other storage devices, personal digital assistants, transmission media, or portable electronic media.
It is acceptable for the SRA to be conducted or reviewed outside the 90-Day MU/EHR reporting period. The SRA or review:
- Must be unique for each EHR reporting period
- Must have a scope including the full EHR reporting period
- Must be conducted within the calendar year of the EHR reporting period
Good to Know: Your EHR vendor may be able to provide information, assistance, and training on the privacy and security aspects of your EHR. However, vendors are not responsible for making their products compliant with HIPAA Privacy and Security Rules
Common Misconceptions: There have been some myths among providers regarding requirements of the SRA for PIP attestation. Common misconceptions include:
- An SRA must be completely redone every year. The truth: A full security risk analysis should be conducted when an EHR is adopted. Each year or when changes to the practice or electronic systems occur, review and update the prior analysis for changes in risks.
- All risks must be mitigated before attestation. The truth: Correcting any deficiencies identified should be completed according to the timeline established in the provider's risk management process.
- SRA's must be outsourced. The truth: It is possible for small practices to do risk analysis themselves using self-help tools. However, doing a thorough and professional risk analysis that will stand up to a compliance review will require expert knowledge that could be obtained through services of an experienced outside professional.
Tips and Available Resources: There is no single method or “best practice” that guarantees compliance, but most risk analysis and risk management processes have steps in common. Here are some considerations as you conduct your risk analysis:
- Review the existing security infrastructure in the practice against legal requirements and industry best practices
- Identify potential threats to patient privacy and security and assesses the impact on the confidentiality, integrity and availability of ePHI
- Prioritize risks based on the severity of their impact on the patients and practice
The HHS Office of the National Coordinator for Health Information Technology (ONC) and the HHS Office for Civil Rights (OCR) updated their Security Risk Assessment Tool in September 2020.The tool is designed for use by small to medium sized health care practices – those with one to 10 health care providers – covered entities, and business associates to help them identify risks and vulnerabilities to ePHI.
In Vermont, Vermont Information Technology Leaders (VITL) offers assistance with security risk assessments, free-of-charge. An analysis tool developed by VITL is used to identify and catalog security issues. A completed analysis is given to the practice which lists the identified vulnerabilities, recommends possible remediation steps, and allows the assessment and rating of risk. Areas requiring a new policy or procedure are also identified. Upon request, VITL can provide policy examples and further guidance with review of identified vulnerabilities and next steps. Some services may require additional costs. Also, please know that VITL is currently doing all SRAs remotely - without a site visit they cannot fully assess or advise on physical security practices and so are currently offering a modified version of the typical security risk assessment. For more information, please contact Mary Donati at mdonati@vitl.net.
Important PY21 Information: Because of the earlier attestation window for PY2021, the requirement to complete an SRA has been modified. Previously, a provider was required to attest to an SRA being completed during the program year for which they are attesting; the SRA or review had to already be completed prior to attestation and documentation of the SRA/review provided with the application.
When providers attest for PY2021, they will be asked if the measure (completing/reviewing an SRA) has been completed prior to the date of attestation. If the response is "no", then they will be asked to attest that the SRA/review will be completed no later than December 31, 2021 and that they understand that their incentive payment will be subject to recoupment for failure to do so.
All attesting providers must upload a copy of their 2021 SRA/review to MAPIR by January 31, 2022, otherwise they will automatically be selected for audit.
PROGRAM YEAR 2021 REPORTING PERIODS (posted 2/18/2021)
There are three separate 90-day time periods to consider when attesting for PIP. They may overlap, or they may be distinct from one another. Here is a quick review of what you need to know about each:
Patient Volume Reporting Period: Each provider must demonstrate patient volume thresholds for each year of program participation. Providers cannot overlap any dates from a previous year's attestation. There are two options for selecting the 90-day period:
- Calendar year preceding payment year (calendar year 2020 currently) OR
- 12 months preceding attestation date (up to 365 days prior to the date you attest in MAPIR).
Click here for additional information on Patient Volume Reporting Periods.
eCQM Reporting Period: For PY2021, the eCQM reporting period is any continuous 90-day period between January 1, 2021 and July 31, 2021. Please note that the attestation window closes on July 31, 2021, so having any reporting period end of this date, while allowed, is not recommended. This reporting period may or may not overlap with either the Patient Volume or the EHR/MU reporting periods.
EHR/MU Reporting Period: For PY2021, the EHR/MU reporting period is any continuous 90-day period between January 1, 2021 and July 31, 2021. Please note that the attestation window closes on July 31, 2021, so having any reporting period end on this date, while allowed, is not recommended. This reporting period may or may not overlap with either the Patient Volume or the eCQM reporting periods. There are measures that can occur before, during, or after the MU reporting period of 90 days (read more here), but within the calendar year of each program year. Additionally, some EHR systems run MU reports for time periods that are slightly more than 90 days (such as 3 month) time periods. If attesting using a reporting period of more than 90 days, a statement explaining the discrepancy should be provided at the time of attestation.
New to PY2021: SRA reporting requirements have changed; please click here for more information.
ATHENAHEALTH UPDATE (posted 2/18/2021)
The Department of Justice released a statement last month announcing that athenahealth will pay a $18.25 million fine after violating the False Claims Act and the Anti-Kickback Statute. It is important to differentiate this example of a violation of the False Claims Act from that of Greenway Health’s violations in 2019. In the Greenway Health violation, the ability of the EHRs’ systems to report for Meaningful Use was directly impacted, which resulted in providers potentially losing incentive payments. In the case of athenahealth, reporting capabilities were not impacted; the issue at hand pertains only to illegal kickback sales process. Customers of athenahealth EHR products should not be negatively impacted when attesting for the Promoting Interoperability Program or similar program.
PY2020 ATTESTATION WINDOW CLOSED (posted 2/5/2021)
Thank you to all the providers and preparers for submitting Program Year 2020 attestations. The Vermont PIP Team is busy reviewing the applications in the order in which they were received. Responding to emails requests for additional information and corrections will allow for faster application processing. We appreciate your cooperation and patience as we complete our process for PY2020 and prepare for PY2021 attestation, which begins April 30, 2021.
AUDIT TIP (posted 2/5/2021)
Question: What happens if I am audited and I fail one Meaningful Use Measure? Would I have to pay back part or all the incentive payment I received for that year?
Answer: Meaningful Use is evaluated in its entirety. If any individual measure cannot be verified or a report shows a measure failed, the full payment must be returned. Payments cannot be broken apart.
STAY TUNED - PY2021 EMAILS COMING SOON (posted 2/5/2021)
The Vermont PIP Team will be sending out emails to providers and organizations that have previously attested to the Promoting Interoperability Program with information about eligibility for Program Year 2021. These emails will include a list of potentially eligible providers as well as the potential amounts of any incentive payments that could be earned through successful attestation. We encourage all recipients to review these emails internally and consider requesting an information webinar to learn more about the program requirements and attestation process.
LAST CALL FOR PY2020 ATTESTATION (posted 1/22/2021)
The Program Year 2020 attestation window closes January 31, 2021. All applications must be completed and submitted in MAPIR by this date to receive an incentive payment. If you have any questions or need support, please email the Vermont Medicaid PIP team at ahs.dvhaEHRIP@vermont.gov.
PROGRAM YEAR 2021 ATTESTATION (posted 1/22/2021)
The current Promoting Interoperability Program sunsets at the end of 2021. As a result, reporting and attestation periods will become truncated to facilitate administration of the program. This will allow for final incentive payments to be paid out by the end of December in 2021. As was the case for PY2020, the attestation window will open during the program reporting year on April 30, 2021 and will run through July 31, 2021. This year is the last year to receive an incentive payment from the Promoting Interoperability Program.
We encourage you to start preparing for attestation by reviewing the PY2021 program requirements, which can be found on the CMS website. Develop a plan now to review your progress, so that any needed clinical workflow adjustments are in place in time for the final 90-day reporting period of the year, which must be completed prior to the date of attestation.
The Vermont PIP Website will be updated with important PY2021 information soon; check back in the coming weeks at https://healthdata.vermont.gov/ehrip
The Vermont Medicaid PIP Team offers web-based consultations to help further your understanding of the PY2021 eligibility, reporting, and documentation requirements. Please email us at ahs.dvhaEHRIP@vermont.gov to schedule a consultation.
PY2021 CQM UPDATES (posted 1/22/2021)
The clinical quality measure (CQM) requirements for Program Year 2021 are:
- Must report on at least 6 CQMs
- Must report on at least 1 outcome measure (if an outcome measure is not available or relevant, then at least 1 high priority measure)
- If no outcome or high priority measures are relevant to a Medicaid EP’s scope of practice, EP may report on any six CQMs that are relevant
CQM reporting period in 2021 is a minimum of any continuous 90-day period within CY 2021, provided that the end date for this period falls before the date of attestation.
HAPPY NEW YEAR! (posted 1/8/2021)
2020 has come to a close (phew!) and many of us are looking forward to a brighter 2021. Even though we are eager to move on, work from 2020 is still not finished. You still have time to attest for Program Year 2020 through January 31, 2021. If you haven’t started yet, we highly recommend you make time next week to start those applications in MAPIR.
Along with the arrival of the new year, the start of PY2021 has begun as well! Attestation for PY2021 looks different because PIP/EHRIP is sunsetting at the end of 2021. The attestation window is from April 30th through July 31st. As a result, the 90-day reporting period must be complete by July 31st, though it is highly recommended that the 2021 reporting period end earlier to give time to complete the application process in MAPIR.
The good news is that program requirements between 2020 and 2021 remain largely unchanged. All objectives and measures will have the same thresholds between these two program years. There are updates to the CQM’s available for reporting, and there are changes to the Security Risk Analysis requirement, which is discussed below. We highly recommend all providers and preparers review program requirements for PY2021 and start preparing for attestation as soon as possible.
SECURITY RISK ASSESSMENTS (posted 1/8/2021)
The Security Risk Assessment (SRA) has been a long-standing requirement for Meaningful Use. In Stage 3, it is a required part of Objective One: Protecting Patients’ Health Information. Because of the earlier attestation window for PY2021, the requirement to complete an SRA has been slightly modified. Previously, a provider had to attest for an SRA being completed during the PY for which they are attesting; the SRA had to already be completed prior to the EP completing the application.
For 2021, an SRA can be completed any time during the calendar year, even after the time period for which a provider has attested. A provider must upload a copy of their SRA to MAPIR by January 31, 2022, otherwise their incentive payment may be recouped.
OBJECTIVE SEVEN, MEASURE THREE (posted 12/23/2020)
Objective Seven in Stage 3 Meaningful Use (MU) includes three measures, all having to do with Health Information Exchange (HIE). Detailed information on this objective can be found here.
Measure Three of this objective has posed challenges for some EP's in the past. As with other objectives, let’s delve into the details of this measure.
For more than 80% of transitions or referrals received and first-time patient encounters, a clinical information reconciliation must be implemented for medications, medication allergies, and current problem list. For more than 80% of transitions or referrals received and first-time patient encounters, a clinical information reconciliation must be implemented for medications, medication allergies, and current problem list.
First encounters with new patients and encounters with existing patients where a summary of care record (of any type) is provided to the receiving EP should be included in the denominator. The summary of care record can be provided either by the patient or by the referring/transiting provider or institution. For patient self-referrals, a patient must disclose the provider from whom they received care in order for that patient to be included in the denominator. If an electronic summary of care record is requested from either the referring provider or the transiting provider and was not received, and the EP queried at least one external source via HIE functionality and could not locate a summary of care, that patient does not have to be included in the denominator.
EP's may exclude from this measure if there are fewer than 100 instances of transitions/referrals/first-time encounters during the reporting period. Also, of the three measures in Objective seven, only two must be met to successfully meet objective requirements.
MULTIPLE LOCATIONS AND EMPLOYERS (posted 12/23/2020)
When attesting to meaningful use (MU), it is important to consider whether or not eligible professionals (EP's) practice at multiple locations and/or have more than one employer. In order to eligible for an incentive payment, at least 50% of an EP’s patient encounters during the reporting period must occur at a location (or a combination of locations) where Certified EHR Technology (CEHRT) is being utilized.
EP’s are required to attest with complete data from all locations equipped with CEHRT to demonstrate meaningful use.
If there are multiple CEHRT’s being used, numerators and denominators from each EHR system should be combined from the systems for attestation in MAPIR.
If MU data from a location cannot be acquired, the denominator must still include all of the patients seen from that location for the reporting period. The numerator may only include patients for whom there is MU data; it is possible to still successfully meet measures but this may inhibit successful performance on some measures.
For additional information, see this PDF from CMS, or email the Vermont PIP Team at ahs.DVHAehrip@vermont.gov
2015 CEHRT REVIEW AND UPDATES (posted 12/11/2020)
CMS and the Office of the National Coordinator for Information Technology (ONC) have established standards and other criteria that electronic health records (EHR’s) must meet in order to qualify for use in the Promoting Interoperability Programs. The latest edition of these standards was created in 2015, hence why the latest requirement for participating in PIP is that providers use 2015 Edition CEHRT (Certified EHR Technology).
For PY2020, 2015 Edition CEHRT does not need to be in place on January 1st of the program year, but functionality does need to be in place on the first day of the reporting period, and the software must meet the criteria for 2015 edition certification by the last day of the reporting period.
ONC’s Cures Act Final Rule made several changes to the existing 2015 Edition certification criteria. The final rule introduced a small number of new certification criteria, revised several existing certification criteria, and it removed several certification criteria. Providers should anticipate that their EHR vendors are making these updates to their software by December 31, 2022. It is highly recommended that providers contact their EHR vendors early and regularly to request updates as soon as they are available. This is important because meaningful use will require this functionality. In past updates, practices have ended up on long waiting lists and have missed out on the opportunity to receive incentive payments because their software did not meet the standards for the reporting period.
For more information about the 2015 Edition CEHRT changes made by the ONC Cures Act, please click here.
AUDIT PRO-TIP (Posted 12/11/2020)
To ensure a smooth audit experience, make sure that multiple people in your organization can retrieve attestation documentation. It is not uncommon that there are staffing changes between the time of attestation and post-payment audit. As a result, we encourage the following:
• Keep supporting documentation in a secure but centralized location
• Share login information for attestation-related programs such as the Vermont Medicaid Portal (needed to access MAPIR) with multiple employees
• Share login information related to any password-protected documents with multiple employees
These practices will ensure that organizations will always have someone who knows where and how to access documentation requested for post-payment audits.
TELEHEALTH AND ATTESTATION (posted 11/25/2020)
The year 2020 has been far from routine for any of us, especially those of us working in the health care industry. Care delivery has changed; technology allowed for providers to connect with patients through virtual means while keeping us safe and distanced whenever possible. The use of telehealth technologies increased greatly during 2020, and it will continue to be a useful tool in health care going forward.
What does the use of telehealth mean for attestation in 2020?
We have heard that some providers believe they may not meet the standards for meaningful use in 2020 because of the increased use of telehealth services. It is important to know that (continued on next page) (continued from previous page) the use of telehealth itself is compatible with meeting MU standards; providers can practice virtually and still successfully attest to meaningful use in PY2020.
However, if operational changes were made as a result of the use of telehealth visits, attestation may be more challenging in some cases. For instance, if documentation or practices that would be required for attestation were skipped as part of a new workflow created to accommodate the use of telehealth, then objectives and measures may not meet the thresholds for meaningful use. In these instances, we encourage preparers to look for a 90-day period when telehealth visits were lowest; perhaps the first quarter of 2020 before operations were changed by the pandemic. Consider when the office might have re-opened for more in-person visits after the first wave of the pandemic as well.
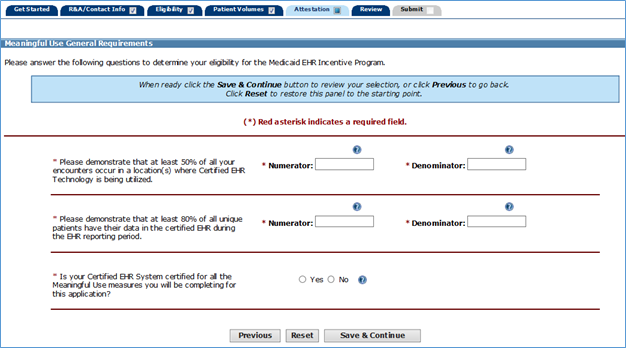
MU GENERAL REQUIREMENTS (posted 11/25/2020)
As part of attestation in MAPIR, preparers will be asked to demonstrate significant use of CEHRT by answering two different eligibility questions (see below). The first question is asking for a percentage of all encounters. The second question is asking for a percentage of unique patients.
For example: A provider has sees 90 unique patients during a reporting period. 75 of them she only sees for one encounter. But there are 15 of them that she sees twice. Adding up the encounters would be (75 + [15*2]) = 105 encounters.
In this case, the denominator in the first question would be 105. The denominator in the second question would be 90. The numerators for each of these would be dependent upon how many encounters (for the first question) or unique patients (for the second question) met the measure outlined in the respective question.
It is highly unlikely that the denominators in these two questions would match, as it is unlikely that a provider would encounter each unique patient exactly one time during a reporting period. Even if 100% of all encounter occurred in locations where CEHRT is being utilized and 100% of all unique patients have their data in the CEHRT, the two denominators would still not be the same.
A CLOSER LOOK: OBJECTIVE EIGHT, PUBLIC HEALTH AND CLINICAL DATA REGISTRY REPORTING (posted 11/13/2020)
There are several measures in this objective that must be answered correctly in order to successfully attest. Please click here for details.
An EP must satisfy two measures for this objective. If the EP cannot satisfy at least two measures, they may take exclusions from all measures they cannot meet.
Note that there are measures 1 through 5. Measures 4 and 5 can be separated into 4a and 4b, and 5a and 5b, respectively, essentially making it possible to attest to a total of seven (7) measures for Objective 8. If an EP attests to active engagement in measure 4a, they must also attest to 4b (which may be satisfied or excluded). Likewise, if an EP satisfies measure 5a, they must also attest to 5b (which may be satisfied or excluded).
Here are some considerations for Vermont EP’s:
- The Blueprint for Health Registry was not active for PY2020. You may not attest to active engagement with this registry for any measure.
- The Vermont Department of Health registries do not meet Stage 3 requirements. You may not attest to active engagement with any of these for any measure, including measures 4 and 5.
- Measures 1, 2, and 3: All Vermont EP's must exclude from these measures. Documentation is not required. Measure 4a: Most EP's will EXCLUDE. However, some EP's may belong to organizations/registries that qualify as an MU Stage 3 Public Health Registry. You must provide documentation for this measure whether you are engaged with a registry or excluding from the measure.
- Measure 4b: If you excluded from 4a, no data entry will be allowed for this measure, therefore no documentation is required. However, if you attested to active engagement in 4a, you must attest to 4b. You must provide documentation for 4b whether you are engaged with a registry or excluding from the measure.
- Measure 5a: Most EP's will EXCLUDE. However, some EP's may belong to organizations/registries that qualify as an MU Stage 3 Clinical Data Registry. You must provide documentation for this measure whether you are engaged with a registry or excluding from the measure.
- Measure 5b: If you excluded from 5a, no data entry will be allowed for this measure, therefore no documentation is required. However, if you attested to active engagement in 5a, you must attest to 5b. You must provide documentation for 5b whether you are engaged with a registry or excluding from the measure.
For samples of the types of acceptable documentation for measures 4a, 4b, 5a, and 5b, please see our Public Health webpage here. You’ll also find information for New Hampshire providers as well as documentation aids for Objective Eight.
PROGRAM YEAR 2021: WHAT TO EXPECT (Posted 10/30/2020)
CMS has released spec sheets for PY2021, the final year to attest for the Medicaid Promoting Interoperability Program. The requirements of the program for PY2021 will remain unchanged from those of PY2020.
While the eCQMs will be updated, the objectives, measures, and exclusions will remain unchanged for the program's final year.
The attestation window for PY2021 is only months away! It will be open from April 30, 2021 through
July 31, 2021. All incentive payments will be distributed by December 31, 2021.
A CLOSER LOOK: OBJECTIVE SIX, MEASURE THREE (Posted 10/30/2020)
Objective Six, Coordination of Care through Patient Engagement, is an objective with which there have been challenges historically, especially with Measure Three. It is worth reviewing this measure in detail to ensure that EPs can attest successfully. Full details can be viewed here.
Objective Six requirements: An EP must attest to all three measures and meet the threshold for two measures. If the EP meets the criteria for exclusions for two measures, they must meet the threshold for the one remaining measure. If the EP meets the criteria for exclusion from all three measures, they may be excluded from meeting this objective.
Measure Three: Patient generated health data or data from a non-clinical setting is incorporated into the CEHRT for more than 5 percent of all unique patients seen by the EP during the EHR reporting period.
Definition for Patient Generated Health Data: Data generated by a patient or a patient's authorized representative.
Definition for Data from a Non-Clinical Setting: This includes, but is not limited to, social service data, data generated by a patient or a patient's authorized representative, advance directives, medical device data, home health monitoring data, and fitness monitor data. The data may not be information the patient provides to the EP during the office visit.
The sources of data may include mobile applications for tracking health and nutrition, home health devices with tracking capabilities such as scales and blood pressure monitors, wearable devices such as activity trackers or heart monitors, patient-reported outcome data, and other methods of input for patient and non-clinical setting generated health data. Telehealth platform, personal health records, social determinants of health screening modules, long term care/post-acute care coordination platforms might also be considered.
Providers in non-clinical settings may include care providers such as nutritionists, physical
therapists, occupational therapists, psychologists, and home health care providers. Other key
providers in the care team such as behavioral health care providers, may also be included.
EXCLUSIONS (Posted 10/16/2020)
Did you know that many objectives and measures for the Promoting Interoperability Program have exclusions that may allow for providers to successfully attest and receive incentive payments, even if they cannot meet all the measures? For instance, if your practice writes less than 100 radiology orders during the reporting period, then you may exclude from the measure associated with electronic radiology orders.
There are also objectives with multiple measures for which all measures do not have to be met to pass the objective. For instance, Objective 7 has three masures to which an EP must attest, but only two of the three measures must be met to pass the objective. And if an EP meets the exclusions for all three measures, the EP can exclude from the entire objective.
For more details about each objective, measures, and exclusions, review the spec sheets from CMS, which can be found here.
SURVEY PARTICIPATION REQUESTED (Posted 10/1/2020)
The Vermont Medicaid PIP team is requesting your participation in a very brief survey in an effort better understand the current environment for participation in PY2020. It will only take a few minutes of your time and is completely anonymous. Click here to access the survey. Thank you for your time!
START OF LAST REPORTING PERIOD FOR PY2020 (Posted 10/1/2020)
The concluding 90-day period of 2020 starts October 3, 2020. This is the final opportunity to ensure you are meeting all MU measures for PY2020.
EHR REQUIREMENTS (Posted 10/1/2020)
you are attesting for PY2020, you MUST be using a 2015 Edition Certified EHR System, also known as a 2015 Edition CEHRT. While the CEHRT does not need to have been in place on January 1, 2020 in order to attest, it must be in place for the entirety of the selected MU reporting period and the product must be certified to the 2015 Edition criteria by the last day of the EHR reporting period.
How can you tell if your EHR system is 2015 Edition CEHRT?
There are a few ways to check:
1. The easiest way is to look at the CEHRT ID, which may or may not be readily available directly within your EHR system. It will be a 15-alpha/numerical code. “15E” will be in the 3rd-5th spaces in the code if your EHR is 2015 Edition.
2. Contact your EHR vendor and ask about the certification standard of your system.
3. Utilize the Certified Health IT Product List (CHPL): Here you can search by product name, vendor name, or CHPL product number to identify the CEHRT ID.
An archive of previously issued VT Medicaid PIP/EHRIP Announcements is available here.
For questions, contact the Vermont EHRIP Team at ahs.dvhaEHRIP@vermont.gov
Back to the Vermont Medicaid PIP/EHRIP Home Page
(Page last updated 07/22/2021)
